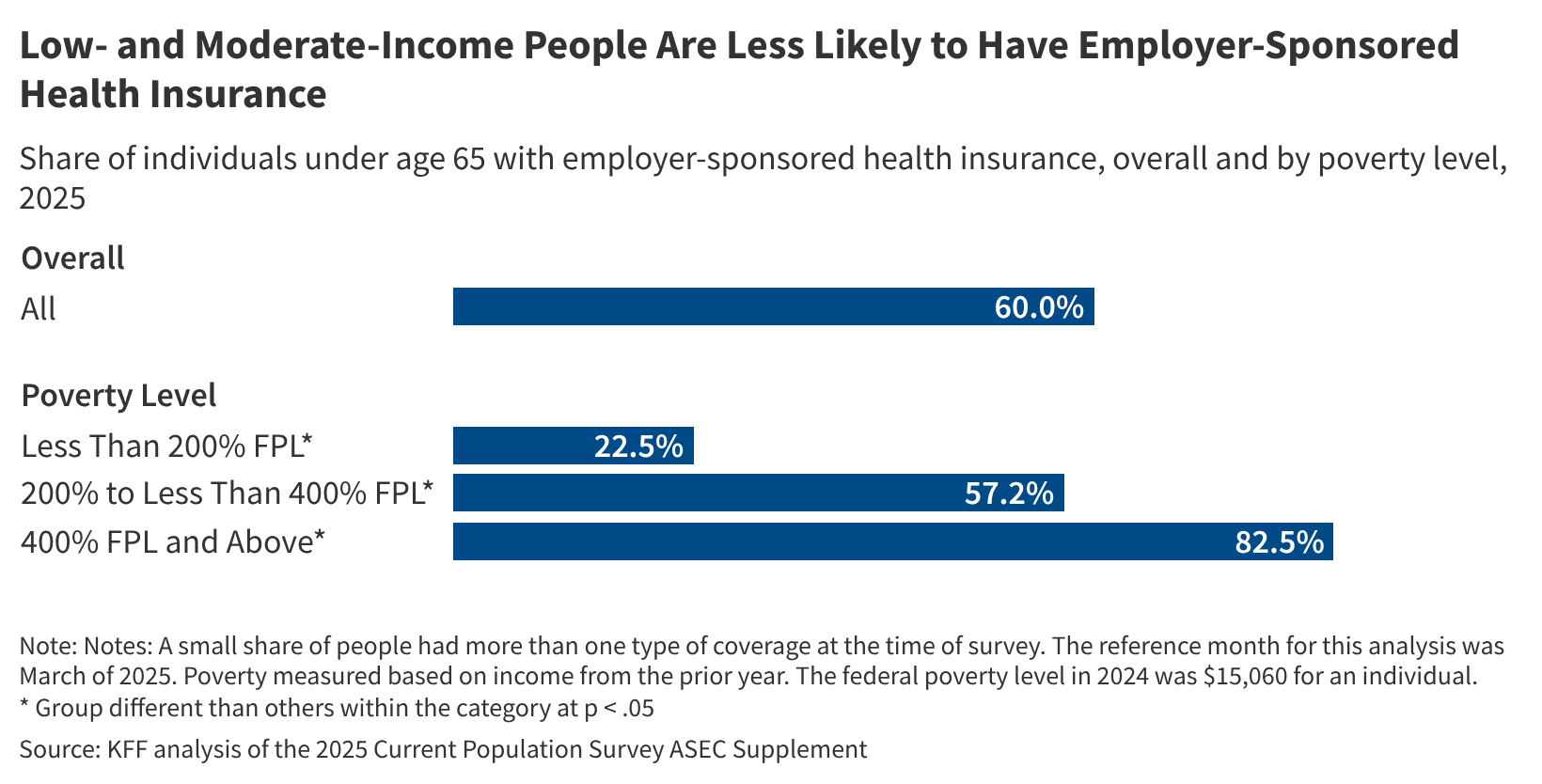Hospital and pharma groups have sharpened their spears for the rematch on 340B rebates.
In comment letters submitted just ahead of a Monday deadline, the stakeholders weighed in on a contentious Trump administration pilot program that would overhaul drug discounts hospitals have received for the past 30 years to subsidize safety-net care.
Rather than receive the discounts at the point of procurement, participating hospitals would need to submit rebates with drugmakers who would then dole out the discount as a rebate. The pharmaceutical industry has said such an approach is necessary to avoid duplicate discounts and diversion amid the subsidy program’s substantial growth—justification the hospital industry has said is unfounded and a cover for adding friction to the process in order to limit payouts.
Much of the feedback received by the Health Resources and Services Administration (HRSA) Monday is treading familiar ground, as the administration’s first attempt at the rebate pilot program was set to begin on Jan. 1 but blocked by hospitals in court on largely administrative grounds. HRSA ultimately pulled its guidance on version one of the 340B Rebate Model Pilot Program, and restarted the process in February with a new round of stakeholder feedback to address a judge’s determination of a rushed process.
However, the latest round of comments highlights some of HRSA’s proposed changes to the pilot’s implementation, attestations made by the government during the prior lawsuit and fresh barbs between sectors locked in a fight over billions of net revenue.
For starters, the American Hospital Association wrote in its April 20 comment letter that HRSA’s February guidance suggests it’s considering an expansion of the pilot’s scope—from version one’s 10 drugs from eight manufacturers to as many as 25 from 13 companies. Accordingly, HRSA’s prior estimate that compliance would require covered entities to devote two additional hours of work per week has been expanded to five additional hours per entity per week. The former reflected a collective $200 million administrative burden on hospitals, while the latter, AHA calculated, would come in around half a billion.
“The previous $200 million labor cost could not justify HRSA’s original Rebate Program; a $500 million one certainly cannot either,” the hospital lobby wrote. “To make matters worse … hospitals have made clear that they will need to devote far more than two to five hours per week to comply with a rebate mechanism.
“…The total administrative costs to 340B hospitals alone will exceed $1 billion. And this figure does not even include other sizable financial expenses like ‘float costs’ and loss off cost-of-goods-sold discounts,” the comment letter reads.
The AHA’s letter also looked to use HRSA’s statements to the court in December against it. At the time, the agency said it was pursuing the rebate model as a way to “balance the interests of two industries at loggerheads.”
The AHA said such a belief “is mistaken,” contrasting the “razor-thin margins” of some safety-net providers against “some of the world’s most profitable publicly traded companies [drugmakers]” as an imbalance. It also said statute instructs HRSA to “give primacy to covered entities,” whereas proposed guidance outlines potential benefits to manufacturers as justification to pursue the pilot.”
“Because there is no sound or lawful reason to abandon the upfront discount model, and because the costs of any switch will massively outweigh any expected benefits, HRSA should not proceed with any rebate mechanism,” AHA wrote. “Any other decision would do serious, irreparable harm to 340B hospitals and the patients they serve.”
An April 20 comment letter from PhRMA, the trade group representing drug manufacturers, largely argued the opposite, urging HRSA to adopt the rebate model so that payers, drugmakers and the public “are no longer obligated to shoulder the ever increasing costs of rampant 340B program violations.”
PhRMA said it was “alarmed and disappointed” when the initial rebate pilot “could not launch following last-minute litigation brought by hospitals adverse to transparency,” and said the decision left drugmakers with no way to prevent duplicate 340B and Medicaid discounts.
The trade group called for rebates to begin “no later than January 1, 2027,” and for the approach to be employed for all 340B drugs “without first limiting implementation to a pilot.” It contested hospital industry claims that the rebate system previously floated by HRSA would impact hospitals’ cash flow, since “they would typically receive rebates well before wholesaler invoices are due,” and stressed the need for mandatory covered entity participation without carveouts.
“We also emphasize that—despite unfounded comments from covered entities—there is no reason to expect the rebate model will affect patients in any way,” PhRMA wrote. “The steep 340B discounts covered entities receive generally are not passed on to patients.
“…The industry strongly supports any efforts to require covered entities to pass through discounts on 340B medicines to uninsured and low-income patients, along with other patient-centered program reforms. A rebate model could directly benefit patients by encouraging 340B patient identification at the pharmacy or point of administration, which is necessary to operationalize passing through discounts to eligible patients,” the letter reads.
Annual total spending on drugs through the 340B program has grown from $6.6 billion in 2010 to $43.9 billion in 2021, or an average of 19% per year, according to a recent analysis from the nonpartisan Congressional Budget Office. The rapid growth has earned scrutiny from some lawmakers and a sympathetic ear from the current administration.
Chief among critics’ claims is that a subset of large health systems, rather than struggling safety-net hospitals, are using the program to pad their margins. Just in Minnesota, where first-in-the-nation transparency requirements provide a unique look at hospitals’ use of the discount program, 181 covered entities received at least $1.34 billion of net 340B revenue. Among them, 12% together collected $1.08 billion and one hospital claimed over a quarter of the total.
The rebate program is likely the most prominent and immediately impactful front in the lobbying and legal war over the 340B program, though several other disputes could lead to policy change.
Earlier this year Eli Lilly and Company and Novo Nordisk began requiring covered entities to provide more data in order to receive discounts, to the protest of hospital group. More recently AbbVie filed a lawsuit challenging the “outdated” definition of a patient under the program, which it said would help limit health system diversions.
All the while a slew of lawsuits are challenging state laws blocking various restrictions on how covered entities contract with outside pharmacies to dispense the drugs to patients. Recent amicus briefs from the federal government, on the side of challenging drugmakers, pushes the scales in their favor and increases the likelihood these cases could reach the Supreme Court.
Publisher: Source link